Published online 2015 May 9. doi: 10.1016/j.sjbs.2015.04.016
PMID: 26858552
This article has been cited by other articles in PMC.
Abstract
Feb 27, 2019 - Urethral catheters are the most commonly deployed medical devices and used to manage a wide range of conditions in both hospital. Keywords- Bacterial Biofilm, Urinary Catheters, E.coli, Drug Resistance. 30% of biofilm forming bacteria are isolated from indwelling medical devices such as.
Self-reproducing microbial biofilm community mainly involved in the contamination of indwelling medical devices including catheters play a vital role in nosocomial infections. The catheter-associated urinary tract infection (CA-UTI) causative Staphylococcus aureus, Enterobacter faecalis, and Pseudomonas aeruginosa were selectively isolated, their phenotypic as well as genotypic biofilm formation, production and monomeric sugar composition of EPS as well as sugar, salt, pH and temperature influence on their in vitro biofilm formation were determined. From 50 culture positive urinary catheters S. aureus (24%), P. aeruginosa (18%), E. faecalis (14%) and others (44%) were isolated. The performed assays revealed their varying biofilm forming ability. The isolated S. aureus ica, E. faecalis esp, and P. aeruginosa cup A gene sequencing and phylogenetic analysis showed their close branching and genetic relationship. The analyzed sugar, salt, pH, and temperature showed that the degree of CA-UTI isolates biofilm formation is an environmentally sensitive process. EPS monosaccharide HPLC analysis showed the presence of neutral sugars (ng/μl) as follows: glucose (P. aeruginosa: 44.275; E. faecalis: 4.23), lactose (P. aeruginosa: 7.29), mannitol (P. aeruginosa: 2.53; S. aureus: 2.62; E. faecalis: 2.054) and maltose (E. faecalis: 7.0042) revealing species-specific presence and variation. This study may have potential clinical relevance for the easy diagnosis and management of CA-UTI.
Keywords: Biofilm, CA-UTI, Exopolysaccharide, Environmental factors, HPLC, SEM
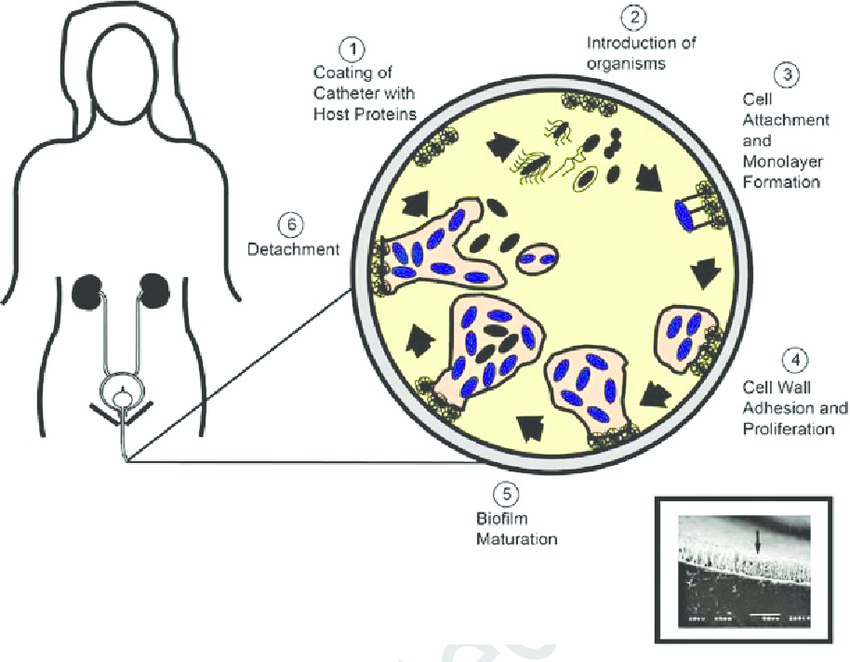
1. Introduction
Healthcare associated infections have emerged as an important ground of morbidity and mortality among hospitalized patients, visitors, and staff. An estimate carried out during the year 2002 accounts that these nosocomial infections cost about $6.7 G (billion) in the United States and £1.06 G (around $1.7G) in the United Kingdom per year (Graves, 2004). The regular occurrence of nosocomial infections is often associated in contact with biomaterials like heart valves, artificial veins, joint prostheses and urinary tract catheters (Dohnt et al., 2011) which are implicated as some of the highly significant risk factors. Among the hospital acquired infections, catheter-associated urinary tract infection (CA-UTI) is the most common one. The United States Centers for Disease Control and Prevention alone records nearly 560,000 CA-UTIs. Biofilm formation is central to CA-UTI pathogenesis (Desai et al., 2010). This biofilm formation is a major facet in nosocomial infections in which soon after the attachment to surfaces pathogens colonize and form a sessile biofilm community, which can develop both on abiotic and biotic surfaces. These biofilms play an imperative role in pathogen physiology, persistence besides serving as a source of various infections. It also acts as an adhesive foundation, defense barrier that protects the embedded cells against detachment by flow shear (Jung et al., 2013). Hence, once became mature, this biofilm is recalcitrant to clearance by both the host immune response and antimicrobial therapies (Brady et al., 2011). Also, the biofilm and their counterpart planktonic cells contrast considerably in their physiology, gene expression pattern, and even morphology. Since they are less sensitive to antimicrobial agents, controlling their growth could be significantly challenging once they are formed (Landini et al., 2010). Furthermore, this biofilm lifestyle’s associated exogenous stress high tolerance, ineffectiveness to antibiotics or other biocide treatments in their eradication (Rendueles et al., 2013) makes use of antibiotics or other antimicrobial agents against a biofilm infection unproductive.
Generally biofilm is made up of non-randomly distributed microcolonies of bacterial cells (15–20% by volume) in a shaped matrix or glycocalyx, the extracellular polymeric substances (EPS) 75–80% by volume (Saini et al., 2011). Hence, the EPS are made up mainly of polysaccharides besides proteins, nucleic acids, lipids, etc., production is the paramount event during biofilm development. The clinical significance of biofilms is their low antimicrobial sensitivity while displaying enhanced pathogenicity (pathogenic synergism). Bacteria living in biofilms exhibit 100- to 1000-fold increase in their antibiotic tolerance in comparison to their free-swimming counterparts (Moscoso et al., 2009, Dufour et al., 2012). The antibiotic resistant common mechanisms including efflux pumps, modifying enzymes, and target mutations could not be accounted for the bacterial protection in the biofilm. The possibility of slow or incomplete penetration of the antibiotics, alternation of chemical microenvironment within the biofilm and formation of a subpopulation having unique, and highly protected, phenotypic cell differentiation similar to spore formation are speculated or assumed as possible reasons (Stewart and Costerton, 2001). In addition, the matrix trapped and concentrated extracellular enzymes such as β-lactamases, formaldehyde lyase, and formaldehyde dehydrogenase inactivates susceptible, typically positively charged, hydrophilic antibiotics (Socransky and Haffajee, 2002). It is proposed that EPS can interact with antibiotics in a manner leading to a decline in their antibacterial activity (Tetz et al., 2009). Sometimes the biofilm matrix acts as an ion-exchange resin so that the strongly charged or highly chemically reactive agents are unable to reach the biofilm’s deeper zones (Socransky and Haffajee, 2002). Hence, antimicrobials express restricted ability to eradicate bacteria deep into biofilms, in part owing to their binding with the biofilm outer layer components (ten Cate, 2012). The association of molecules like EPS and DNA within the biofilm constitutes a physical barrier to the diffusion of antimicrobial agents (Bordi and de Bentzmann, 2011). Consequently, the chemotherapeutic agents find difficulty in penetrating the polysaccharide matrix to reach and affect the microorganisms. So, the matrix helps to increase the chances of the colonies’ survival by protecting bacteria deep inside the biofilm from antibiotics and antiseptics (Gurenlian, 2007). Hence manifestation of these characters that make sessile microorganisms more resistance to antimicrobial agents than their planktonic counterparts is the highly human being damaging property of biofilms (Villa and Cappitelli, 2013).
The principal component of EPS determines the physical properties of the biofilm while the bacterial cells determine its physiological properties (De Beer and Stoodley, 2006). They are accountable for most of their physical, chemical, and biological properties (Ruzicka et al., 2011). The occurrence of different types of polysaccharides and their production is the species and strain dependent one. The Pseudomonas aeruginosa alginate, staphylococcal polysaccharide intercellular adhesion, streptococcal and lactobacilli glucans and fructans are examples of best-known biofilm-associated EPS (Ruzicka et al., 2011). They act as a backbone of the biofilm by binding the biofilms bacteria together in a sticky web of tangled EPS fibers which connect cells as well as anchor them to a surface and to each other (Sihorkar and Vyas, 2001). They play an important role in attachment, detachment, mechanical strength, antibiotic resistance exo-enzymatic degradation activities (De Beer and Stoodley, 2006), cell-to-cell interconnection, interactions between subpopulations, tolerance, and exchange of genetic material (Harmsen et al., 2010). Therefore, EPS plays a vital role not only in their formation but also behavior (Ruzicka et al., 2011). Hence, they are deemed to be necessary for biofilm lifestyle existence and global expression of bacterial pathogen’s virulence (Xiao et al., 2012).
There are well-described differences in the production of these polysaccharides (Ruzicka et al., 2011) which may vary in chemical and physical properties (Dufour et al., 2012). Environmental factors are the underlying truth in the establishment of biofilms particularly in abiotic surfaces especially in foreign devices and surgical equipment (Hess et al., 2011). Numbers of environmental parameters have an effect on the partitioning of cells between planktonic and biofilm phases and hence influences the successful establishment of biofilms in medical, industrial and environmental settings. Recently it is suggested that the environmental signals regulate the initiation of species-specific biofilm thereby allowing efficient colonization of each bacterial species to its preferred environment (Rinaudi et al., 2006). Though the environmental conditions are found to influence the EPS production and biofilm formation, their mechanisms have not been clearly defined until now (Jung et al., 2013). There is a dearth of data in the literature regarding to which, and what extent the diverse environmental parameters affect the biofilm build-up of these CA-UTI organisms which is paramount since they form a biofilm outside the host organism. It is also necessary to measure the impact of environmental parameters on the pathogen EPS production directly by biofilm specific methods in order to determine their in vitro biofilm formation on indwelling medical devices. However, the current methods make use of biofilm thickness and/or density, microscopic coupled viable cell counting techniques like indirect proxies, which are not, only inaccurate and/or not amenable to high-throughput technologies. Direct measuring of some component of the exopolysaccharide matrix would be more useful and productive (Mojica et al., 2007). Further the effects of temperature, sugar, salt, relative humidity levels, and nutrient composition like environmental factors on biofilm production, EPS composition and colony establishment have not been well established in urinary catheter implicated urinary tract infection. Thus, we investigated the in vitro biofilm forming and EPS producing ability of the CA-UTI clinical isolates, their EPS monomeric sugar composition and the influence of environmental parameters on them. We believe that understanding the nature of biofilm and their components as well as, their possible linkage with the environmental factors would help in CA-UTI effective treatment and management.
2. Materials and methods
2.1. Sample processing
Urinary catheter samples were collected from government headquarters hospital Erode district, Erode, Tamilnadu, India during a two year collection period from June 2009 to June 2011 with proper care. The urinary catheter samples removed from patients diagnosed with bacteremia showing positive urine culture with >105 CFU/mL 48 h after admission due to CA-UTI were collected. The collected catheter samples were transported to the routine diagnostic laboratory within 2 h, inoculated into Luria Bertani broth aseptically and incubated overnight at 37 °C. The growth of the organisms were dispersed, inoculated onto selective media like mannitol salt agar, MacConkey agar, Enterococcus selective agar, cetrimide agar and blood agar (HiMedia, Mumbai, India) for selective isolation of S. aureus, E. faecalis and P. aeruginosa. The obtained isolates were identified by colony morphology; Gram’s staining and was characterized biochemically following standard methods (Cappuccino and Sherman, 1996, Monica Chesbrough, 1998).
2.2. Assessment of adherence and biofilm forming ability of the CA-UTI isolates
The CA-UTI bacterial isolate’s adherence, biofilm formation, slime and EPS production as well as their quantification were determined by the following three different methods.
2.2.1. Christensen test tube method
The qualitative method of Christensen et al. (1982) was employed for determining the adherence and biofilm forming ability of the isolates. Briefly, a loop full of the bacterial culture (each isolates) from the agar plate was inoculated into a sterile glass test tube containing 5 ml of trypticase soy broth (TSB, HiMedia) and incubated at 37 °C for 48 h. Each tube was decanted, stained with 0.25% safranin or crystal violet, gently rotated to ensure uniform staining, and then the contents were decanted gently. The tubes were then placed upside-down to ensure draining. The tube’s inner surface color was observed and used for interpretation. An adherent film on the surface of the glass tube was taken as an evidence of slime formation. The absence of a film represented as a negative result (−). Based on slime production, the positive results were recorded as strong (+++), moderate (++), and weak (+) (Murugan et al., 2010). Two different observers interpreted each test.
2.2.2. Congo red agar (CRA) method
Slime productions by each bacterial isolate were determined by the CRA method as described (Freeman et al., 1989). CRA plates were streaked with each bacterial isolate and incubated aerobically for 24 h at 37 °C. The appearance of dark-black colonies with a rough, dry, and crystalline consistency was considered as indicative of slime production. Non-slime isolates produced pinkish red smooth colonies with a darkening at the center.
2.2.3. Microtiter plate analysis
The isolate’s in vitro biofilm forming ability was quantified using the microtiter plate analysis as described (Tendolkar et al., 2004, Murugan et al., 2011). Briefly, all the bacterial isolates were individually grown overnight in TSB at 37 °C and diluted (1:40) with the same broth supplemented with 0.75% glucose. The cells were pelleted, suspended in a fresh medium and normalized to an absorbance of 1.00 at 595 nm. The cultures were diluted 1:40 with the same broth and 200 μL of the same was dispensed into wells of a flat-bottomed polystyrene microtiter plate (Tarsons, Mumbai, India). After 48 h of static incubation at 37 °C, the wells were decanted, washed three times with phosphate-buffered saline (PBS) to remove planktonic and loosely bound cells and air-dried at room temperature. The adherent bacteria were stained with 150 μL of 0.1% crystal violet for 15 min. After rinsing twice with 200 μL of sterile phosphate buffer saline and drying for 5 min at room temperature, light absorbance was measured at 490 nm using a microtiter reader (model 680, Bio-Rad, Hercules, CA). A three-grade scale was used to evaluate the strain’s slime producing ability based on OD as negative (<0.500); + (0.500 <1.500); ++ (>1.500) as scored (Alcaaz et al., 2003).
2.3. PCR amplification of biofilm encoding genes
The presence of respective biofilm encoding genes; ica gene in S. aureus, esp gene in E. faecalis and cup A gene in P. aeruginosa were searched among the isolates. These gene loci were taken from GenBank sequence database of the National Center for Biotechnology Information (http://www.ncbi.nlm.nih.gov). The primers designed and used for polymerase chain reaction (PCR, Eppendorf, Hamburg, Germany) amplification of virulence genes includes (1) ica gene forward -5′- AAGTCATACACTTGCTGGCG-3′ and reverse -5′-CTGTCTGGGCTTCACCATGT-3′ (Murugan et al., 2010); (2) esp gene forward -5′- TTTTGGGGCAACTGGAATAG- 3′and reverse -5′-TTCTGCCCCAGCAAATAGTC-3′; (3) cup A- gene forward -5′ AATTCGATGATCGCCTGTT-3′ and reverse -5′GCGATAGAGGTTGGTGTCGT-3′. Genomic DNA from the isolates was isolated and purified using the DNeasy Tissue kit (Qiagen, Valencia, CA). PCR reactions were carried out using Taq polymerase (Invitrogen, Carlsbad, CA) with the following cycle parameters: 95 °C for 2 min followed by 30 cycles at 94 °C for 1 min, 52–63 °C for 1 min (depending on the gene), and 72 °C for 2 min. The cycle ended by an extension for 10 min at 72 °C (Marra et al., 2007). Amplified sequences submitted to NCBI were assigned with accession numbers (HM140703 and HM140704; esp gene of E. faecalis strains KMS E 01 and KMS E 07) and HM 140705 and HM 140706; cup A gene of P. aeruginosa strains KMS P03 and KMS P05). Evolutionary relationship among the isolates was determined by phylogenetic analysis. The sequences were aligned using CLUSTAL W. The resulting distances are used to calculate a phylogenetic guide tree, which uses pairwise sequence distance calculation to perform multiple sequence alignment. The initial CLUSTAL W calculation was found to be a crude similarity measure between all pairs of sequences by using a fast and approximate alignment algorithm (Wilbur and Lipman, 1983) and then the determination of the order sequences to be aligned in the final multiple alignment. The guide tree is calculated using MEGA 4.
2.4. Effect of environmental parameters on biofilm formation
To determinate the effect of NaCl, about 200 μL of TSB was dispensed in each row of the microtiter plate. The first set of plate wells was incorporated with NaCl in increasing concentrations from 0.1 mg/mL to 1 mg/mL along with 200 μL of TSB. Then each well was inoculated with 20 μL of respective selected cultures, covered and incubated aseptically at 37 °C for 48 h. After incubation, wells were emptied and stained, destained as described (Pitts et al., 2003). Formed biofilm was quantified in duplicate by a microtiter plate reader (Bio-Rad, Hercules, CA) at 490 nm with control wells. The above procedure was repeated for sugar at concentrations ranging from 1% to 8%. Effect of pH on the biofilm forming ability of the isolates was determined by adjusting the culture medium pH from 2 to 13 using 0.1 M NaOH and 0.1 M HCl. To study the impact of temperature, inoculated broth containing plates were incubated at 10 °C, 20 °C, 30 °C, 40 °C, 50 °C and 60 °C with all the isolates. From the obtained OD values, correlation coefficient, the coefficient of variation was calculated to determine the influence of parameters on biofilm formation.
2.5. EPS extraction and quantification
The selected CA-UTI isolates were grown in a basal salt solution (BSS) containing (g/L): NaCl 30.0; KCl 0.75; MgSO4 7H2O 7.0; NH4Cl 1.0; K2HPO4 0.7; KH2PO4 0.3; glucose 10.0 and 1 mL of trace metal solution. Determination of the effects of nutrients on EPS, their extraction and quantification were carried out as described (Majumdar et al., 1999). Carbon and nitrogen sources and concentrations of phosphate were varied as required and pH of the medium was adjusted to 7.5. Bacterial cells were pelleted by centrifugation at 10,000 rpm for 30 min. To the supernatant, 2.2 volumes of absolute chilled ethanol were added to precipitate EPS from S. aureus and E. faecalis. For P. aeruginosa concentrated KCl 25% (w/v) was added attaining a final concentration of 1% (w/v) to aid the precipitation and 2–3 volumes of isopropanol was added to precipitate the EPS after overnight incubation at 4 °C. The precipitated EPS was collected as pellets by centrifugation quantified by weighing and lyophilized.
2.6. HPLC EPS monomeric sugar analysis
The lyophilized EPS was analyzed for its monomeric sugar composition by using the Agilent 1200 HPLC system (Agilent Technologies Inc., Santa Clara, CA, USA) equipped with Meta-Crap 87C column and a refractive index detector (1.00–1.75 RIU). 8 μL sample was injected using a quart pump into the system that uses double sterile distilled water as the mobile phase. The flow rate was adjusted to 0.6 mL/min, and 80 °C was maintained. Sugars were identified after hydrolyzing the EPS with methanol by a refractive index detector based on the peak value. The sugar concentration was calculated by the following formula.
Concentration of sugar = Amount of sugar in 200 μL of sample/Weight of the sample.
3. Results
To evaluate the CA-UTI pathogen biofilm forming ability and their response to environmental influence, we chose to isolate the biofilm forming CA-UTI causatives from the catheters removed from infected patients. A total of 50 catheter samples were collected from UTI and diagnosed from randomly selected 22–65 year-old (32 male and 18 female) patients. All the catheter samples collected were found to be culture positive. The individual isolates obtained from the selective media plates were presumptively identified using conventional microbiological methods including both culture-based methods and biochemical tests respectively. Although sampled from identically diagnosed infections, the isolates showed varied frequencies of isolation. It includes S. aureus (24%), P. aeruginosa (18%) and E. faecalis (14%), and others (44%).
They were further analyzed for their biofilm forming as well as slime producing ability and are graded accordingly. The bacterial surface attachment, biofilm build-up and responsible slime producing ability of the isolated urogenic organisms were determined using tube adherence test, CRA plate method and confirmed using the microtitre plate assay. Considerable variation was observed between the CA-UTI in their biofilm ability. Christensen tube adherence test was performed where the growth and color intensity of the inner surfaces of the tubes were observed, 58% of the isolates formed a continuous film of EPS on the inner surface of the tube. They were considered as strong slime producers, 26% were categorized as moderate, and 16% were weak in slime formation. To confirm the biofilm forming abilities of the urogenic isolates, the “colony biofilm” assay in CRA plates was performed which indicated that 48% of the isolates were strong slime formers and 52% were non slime formers. The isolate’s biofilm formation was further assessed and quantified using the micro-titer well plate technique semi-quantitative evaluation. The formation of the biofilm was determined by reading the optical density of the wells at 490 nm using microtiter reader. From the isolate’s optical density readings considered as an indicative of higher activity, the E. faecalis KMS E01, P. aeruginosa KMS P03 and KMS P05, S. aureus strains KMS S01 and KMS S03 were found to be very high biofilm formers according to their group concern which were used for further study.
The strong biofilm formers biofilm promoting esp gene of E. faecalis, ica gene of S. aureus and cup A gene of P. aeruginosa were amplified. E. faecalis KMS E01 esp gene amplicon was found to have 519 bp. The amplicons of cup A gene of P. aeruginosa KMS P03 and KMS P 05 has a length of 448 bp and 345 bp respectively. The S. aureus strains KMS S01 and KMS S03 ica A encoding gene fragment sizes were about 630 bp and 580 bp. Phylogenetic affiliation of the high biofilm forming CA-UTI isolates revealed the similarity between the amplicons of their biofilm encoding genes (Fig. 1). The fingerprints obtained were recorded and subjected to computerized analysis. The isolate’s ica, esp and cup A gene sequences and the other biofilm forming and medical devices indwelling organisms GenBank reference sequences were found to show high genetic relatedness as indicated by their close branching. Sequence similarity also suggests the possibility of biofilm encoding genes horizontal transfer. Both genotypic and phenotypic biofilm forming capability of the isolates along with their exopolysaccharide synthesizing ability revealed the isolates suspected possible role in the initiation and manifestation of CA-UTI.
(a) Showing the phylogenetic relationship between the CA-UTI isolate Staphylococcus aureus KMS01 and others inferred from ica gene. (b) Showing the esp gene inferred phylogenetic relationship between the CA-UTI isolate and other reference database Enterococcus faecalis organisms. (C) Showing the phylogenetic relationship between the CA-UTI isolates Pseudomonas aeruginosa KMS P01 and KMS P05 and others inferred from cup A gene.
The influence of temperature, pH, sugar, and salt concentration on CAUTI biofilm forming isolates in vitro biofilm build-up were also studied. The varying growth temperature effects on the biofilm build-up and EPS given in (Fig. 2) revealed the significance of growth temperature on the CA-UTI isolates biofilm formation The mean quantities of EPS formed at 40 °C (0.81 ± 075) were higher than other temperatures both at high and low levels for the selected isolates whose mean values range from 0.80 ± 0.39 to 1.9 ± 0.78. Biofilm formation positively correlated with temperature (Fig. 2a). The biofilm build-up by CA-UTI isolated E. faecalis increased with increasing pH in the interval of 5.5–6.7, attained maximum at the pH 5.5 (0.656 ± 0.258). P. aeruginosa strains showed a significant amount of EPS formation at the pH 6 (0.647 ± 0.28), whereas the strains of S. aureus exhibited higher EPS at neutral pH 7 (0.597 ± 0.31). When the pH crossed the neutral, there was a gradual decrease in biofilm formation showing the high influence of hydrogen ion concentration on EPS production. Both extremes of pH did not support EPS production. Maximum EPS production fluctuated between pH 6 and pH 8 (Fig. 2b). The figure (Fig. 2c) shows that addition of sugar as carbon source had an increased effect on the biofilm build-up and hence EPS production of all CA-UTI isolates which attained the maximum when the medium was supplemented with 4% of sugar. The production of biofilm increased with an increase in sugar concentration. The figure (Fig. 2d) demonstrates the effect of salt concentration on the biofilm build-up. EPS production got increased along with salt concentration; however, the biofilm build-up decreased above 3.5% of salt concentration. All the four influencing factors, the sugar, salt, pH and temperature have shown a positive correlation with biofilm formation. The coefficient of variation values emphasized the stability of the organisms under the selected parameters. All the isolates showed maximum stability against salt and sugar concentrations but the isolates were affected significantly by temperature and pH variations.
Showing the influence of environmental factors (a) temperature, (b) pH, (c) sugar and (d) salt on EPS production of CA-UTI isolates.
The studied CA-UTI pathogens E. faecalis KMS E01 and KMS E07 were found to produce 174 and 176 mg/L of EPS. The CA-UTI P. aeruginosa isolates KMS P03 and KMS P05 produced 189 and 193 mg/L of EPS respectively, whereas the S. aureus isolates KMS S01 and KMS S03 produced 187 and 183 mg/L in glucose containing medium. The EPS monosaccharide HPLC chromatogram (Fig. 3) revealed the presence of neutral sugars glucose, lactose and mannitol in the P. aeruginosa biofilm at a concentration of 44.275 ng/μL, 7.29 ng/μL and 2.53 ng/μL respectively where glucose dominated other sugars. Mannitol was the major polysaccharide present in the biofilm of S. aureus in the concentration of 2.62 ng/μL. The polysaccharides produced by E. faecalis were composed of glucose (4.23 ng/μL), maltose (7.0042 ng/μL) and mannitol (2.054 ng/μL).
HPLC chromatograms showing monomeric sugar composition of EPS. Pseudomonas aeruginosa EPS showing lactose (a) and manitol (b), Staphylococcus aureus EPS showing manitol (c), Enterobacter faecalis EPS showing glucose (d), manitol (e) and maltose (f).
4. Discussion
The CA-UTI number upsurges every year since urinary catheters are the second most often used and internally placed human body foreign objects through which the causatives more easily attack the urinary tract and urinary bladder (Hola et al., 2012). Biofilms readily form on the inner or outer surfaces of these tubular latex or silicone devices when inserted into the urinary tract in which the biofilm developing tendency of the organism increases with catheterization time (Al-Mathkhury et al., 2011). Catheterization increases the risk of developing bacteriuria about 3–6% per day (Ortega et al., 2013). About 80% of the UTIs in acute-care units are CA-UTIs. Almost 50% of short-term catheterized patients acquire infections within this period whereas the risks of infection from the long-term catheters are 100% (Dohnt et al., 2011). The microbial species causing CA-UTI are different from those causing bacteremic UTI without urological manipulation (Ortega et al., 2013). In the present study, the high biofilm active isolates like S. aureus, P. aeruginosa, and E. faecalis were selectively isolated from the catheters removed from patients suffering from CA-UTI. A number of previous studies had shown the urinary tract colonization and infection by S. aureus in patients with IDUC (indwelling urinary catheters) and a history of instrumentation, surgery, or obstruction of the urinary tract. Also, many studies reported S. aureus as a primary urinary pathogen and its manipulation S. aureus bacteriuria (SABU) as a potential reservoir for invasive infection (Choi et al., 2009). Al-Mathkhury et al. (2011) demonstrated the Gram negative opportunistic P. aeruginosa common colonization of urinary catheters and biofilm development on them. E. faecalis is the 3rd most common hospital pathogen causes ∼12% of nosocomial infections (Marra et al., 2007) and is a significant etiological agent of CA-UTI. It grasps advantage of the foreign body presence in the bladder for producing biofilm thereby establishing persistent urinary tract infections during CA-UTIs. It explains the clinical observation and frequent recovery of E. faecalis from patients with foreign body-associated infections (Guiton et al., 2013) including catheter.
The central character in the story of CA-UTI is the biofilm. They offer survival advantages to the difficult to eradicate urinary catheter biofilm associated microorganisms ensuing persistent antimicrobial resistant infections (Trautner and Darouiche, 2004). All the selected CA-UTI isolates S. aureus, P. aeruginosa, and E. faecalis exhibited high biofilm forming ability. Earlier Abdallah et al. (2011) pointed out the difference between the biofilm forming ability of isolates from CA-UTI (43.3%) patients and UTI without indwelling catheter (30%) patients. All biofilm producing ureteral catheterization staphylococcal isolates were found to be positive for ica genes, which serve as, virulence markers in these staphylococci urinary catheterization infections (Gad et al., 2009).
Exopolysaccharides play a crucial role in the formation of biofilms and biofilm resistance to antimicrobials and innate host defense. The observed behavior of CA-UTI isolates during varying temperature, pH, sugar and salt concentrations revealed the impact of environmental factors on them. It leads to agreement with the views of Ledeboer and Jones (2005) who stated that despite bacterial species possession of genes determining the produced EPS amount and composition environmental surfaces and conditions are also imperative. Though the biofilm mode enables survival under a multiplicity of conditions, the bacteria return to their planktonic state when the environmental conditions change or their microenvironment becomes unfavorable (Amalaradjou and Venkitanarayanan, 2013).
5. Conclusion
Since the surface-associated exopolysaccharides and capsules play a role in both extracellular and intracellular adherence during the conversion from planktonic to biofilm growth, their characterization would help the identification and design of more effective anti-biofilm therapeutic agents (Bales et al., 2013). The obtained result of factors influencing exopolysaccharide formation would be a significant one as far as the CA-UTI is concerned as they will direct us toward predicting the environmental factors predisposing the dispersal of catheter biofilm organisms. Hence, the risk of clinical process as a benign or acceptable side effect of CA-UTI can be easily diagnosed and their ascension can be better controlled by understanding the causatives of biofilm exopolysaccharide component nature and the impact of environmental factors upon them.
Acknowledgment
The authors extend their appreciation to the Deanship of Scientific Research at King Saud University for funding the work through the research group project No. RGP-VPP-183.
Footnotes
Peer review under responsibility of King Saud University.
References
- Abdallah N.M.A., Elsayed S.B., Mostafa M.M.Y., El-Gohary G.M. Biofilm forming bacteria isolated from urinary tract infection, relation to catheterization and susceptibility to antibiotics. Int. J. Biotechnol. Mol. Biol. Res. 2011;2(10):172–178.[Google Scholar]
- Alcaaz L.E., Satorres S.E., Lucero R.M., Centorbi O.N.P. Species identification, slime production and oxacillin susceptibility in coagulase – negative staphylococci isolated from nosocomial specimens. Braz. J. Microbiol. 2003;34:45–51.[Google Scholar]
- Al-Mathkhury H.J.F., Ali A.S., Ghafil J.A. Antagonistic effect of bacteriocin against urinary catheter associated Pseudomonas aeruginosa biofilm. N. Am. J. Med. Sci. 2011;3:367–370.[PMC free article] [PubMed] [Google Scholar]
- Amalaradjou M.A.R., Venkitanarayanan K. Role of bacterial biofilms in catheter-associated urinary tract infections (CAUTI) and strategies for their control. In: Nelius Thomas., editor. InTech; 2013. (Recent Advances in the Field of Urinary Tract Infections). [Google Scholar]
- Bales P.M., Renke E.M., May S.L., Shen Y., Nelson D.C. Purification and characterization of biofilm-associated EPS exopolysaccharides from ESKAPE organisms and other pathogens. PLoS ONE. 2013;8(6):e67950.[PMC free article] [PubMed] [Google Scholar]
- Bordi C., de Bentzmann S. Hacking into bacterial biofilms: a new therapeutic challenge. Ann. Intensive Care. 2011;1:19.[PMC free article] [PubMed] [Google Scholar]
- Brady R.A., O’May G.A., Leid J.G., Prior M.L., Costerton J.W., Shirtliff M.E. Resolution of Staphylococcus aureus biofilm infection using vaccination and antibiotic treatment. Infect. Immun. 2011;79(4):1797–1803.[PMC free article] [PubMed] [Google Scholar]
- Cappuccino J.G., Sherman N. Fifth ed. The Benjamin/Cummings Publishing Company Inc; California: 1996. Microbiology a Laboratory Manual. [Google Scholar]
- Choi S.H., Lee S.O., Choi J.P., Lim S.K., Chung J.W., Choi S.H., Jeong J.Y., Woo J.H., Kim Y.S. The clinical significance of concurrent Staphylococcus aureus bacteriuria in patients with S. aureus bacteremia. J. Infect. 2009;59:37–41. [PubMed] [Google Scholar]
- Christensen G.D., Simpson W.A., Bisno A.L., Beachey E.H. Adherence of slime producing strains of Staphylococcus epidermidis to smooth surfaces. Infect. Immun. 1982;37(1):318–326.[PMC free article] [PubMed] [Google Scholar]
- De Beer D., Stoodley P. Microbial biofilms. In: Dworkin M., Falkow S., Rosenberg E., Schleifer K.H., Stackebrandt E., editors. Prokaryotes. Springer-Verlag; New York: 2006. pp. 904–937. [Google Scholar]
- Desai D.G., Liao K.S., Cevallos M.E., Trautner B.W. Silver or nitrofurazone impregnation of urinary catheters has a minimal effect on uropathogen adherence. J. Urol. 2010;184:2565–2571.[PMC free article] [PubMed] [Google Scholar]
- Dohnt K., Sauer M., Müller M., Atallah K., Weidemann M., Gronemeyer P., Rasch D., Tielen P., Krull R. An in vitro urinary tract catheter system to investigate biofilm development in catheter-associated urinary tract infections. J. Microbiol. Methods. 2011;87:302–308. [PubMed] [Google Scholar]
- Dufour D., Leung V., Levesque C.M. Bacterial biofilm: structure, function, and antimicrobial resistance. Endod. Topics. 2012;22:1538–1601.[Google Scholar]
- Freeman D.J., Falkiner F.R., Keane C.T. New method for detecting slime production by coagulase negative staphylococci. J. Clin. Pathol. 1989;42:872–874.[PMC free article] [PubMed] [Google Scholar]
- Gad G.F., El-Feky M.A., El-Rehewy M.S., Hassan M.A., Abolella H., El-Baky R.M. Detection of icaA, icaD genes and biofilm production by Staphylococcus aureus and Staphylococcus epidermidis isolated from urinary tract catheterized patients. J. Infect. Dev. Countries. 2009;3(5):342–351. [PubMed] [Google Scholar]
- Graves N. Economics of preventing hospital infection. Emerg. Infect. Dis. 2004 Available from: < http://wwwnc.cdc.gov/eid/article/10/4/02-0754.htm>. [Google Scholar]
- Guiton P.S., Hannan T.J., Ford B., Caparon M.G., Hultgren S.J. Enterococcus faecalis overcomes foreign body-mediated inflammation to establish urinary tract infections. Infect. Immun. 2013;81(1):329–339.[PMC free article] [PubMed] [Google Scholar]
- Gurenlian J.A.R. The role of dental plaque biofilm in oral health. J. Dent. Hyg. 2007;81(5):116.[Google Scholar]
- Harmsen M., Yang L., Pamp S.J., Tolker-Nielsen T. An update on Pseudomonas aeruginosa biofilm formation, tolerance, and dispersal. FEMS Immunol. Med. Microbiol. 2010;59(3):253–268. [PubMed] [Google Scholar]
- Hess D.J., Henry-Stanley M.J., Wells C.L. Gentamicin promotes Staphylococcus aureus biofilms on silk suture. J. Surg. Res. 2011;170(2):302–308.[PMC free article] [PubMed] [Google Scholar]
- Hola V., Peroutkova T., Ruzicka F. Virulence factors in Proteus bacteria from biofilm communities of catheter-associated urinary tract infections. FEMS Immunol. Med. Microbiol. 2012;65:343–349. [PubMed] [Google Scholar]
- Jung J.H., Choi N.Y., Lee S.Y. Biofilm formation and exopolysaccharide (EPS) production by Cronobacter sakazakii depending on environmental conditions. Food Microbiol. 2013;34(1):70–80. [PubMed] [Google Scholar]
- Landini P., Antoniani D., Burgess J.G., Nijland R. Molecular mechanisms of compounds affecting bacterial biofilm formation and dispersal. Appl. Microbiol. Biotechnol. 2010;86(3):813–823. [PubMed] [Google Scholar]
- Ledeboer N.A., Jones B.D. Exopolysaccharide sugars contribute to biofilm formation by Salmonella enterica serovar Typhimurium on HEp-2 cells and chicken intestinal epithelium. J. Bacteriol. 2005;189(9):3214–3226.[PMC free article] [PubMed] [Google Scholar]
- Majumdar I., D’souza F., Bhosle N.B. Microbial exopolysaccharides: effect on corrosion and partial chemical characterization. J. Indian Inst. Sci. 1999;79:539–550.[Google Scholar]
- Marra A., Dib-Hajj F., Lamb L., Kaczmarek F., Shang W., Beckius G., Milici A.J., Medina L., Goodtz T.D. Enterococcal virulence determinants may be involved in resistance to clinical therapy. Diagn. Microbiol. Infect. Dis. 2007;58:59–65. [PubMed] [Google Scholar]
- Mojica K., Elsey D., Cooney M.J. Quantitative analysis of biofilm EPS uronic acid content. J. Microbiol. Methods. 2007;71:61–65. [PubMed] [Google Scholar]
- Monica Chesbrough . Cambridge University Press; London: 1998. District Laboratory Practice in Tropical Countries. Vol. I & II. [Google Scholar]
- Moscoso M., García E., Lopez R. Pneumococcal biofilms. Int. Microbiol. 2009;12:77–85. [PubMed] [Google Scholar]
- Murugan K., Usha M., Malathi P., Sohaibani S.A., Chandrasekaran M. Biofilm forming multidrug resistant Staphylococcus spp. among patients with conjunctivitis. Pol. J. Microbiol. 2010;59:233–239. [PubMed] [Google Scholar]
- Murugan K., Selvanayaki K., Sohaibani S.A. Antibiofilm activity of Andrographis paniculata against cystic fibrosis clinical isolate Pseudomonas aeruginosa. World J. Microbiol. Biotechnol. 2011;27:1661–1668.[Google Scholar]
- Ortega M., Marco F., Soriano A., Almela M., Martínez J.A., Pitart C., Mensa J. Epidemiology and prognostic determinants of bacteraemic catheter-acquired urinary tract infection in a single institution from 1991 to 2010. J. Infect. 2013;67(4):282–287. [PubMed] [Google Scholar]
- Pitts B., Hamilton M.A., Zelver N., Stewart P.S. A microtiter – plate screening method for biofilm disinfection and removal. J. Microbiol. Methods. 2003;54:269–276. [PubMed] [Google Scholar]
- Rendueles O., Kaplan J.B., Ghigo J.M. Antibiofilm polysaccharides. Environ. Microbiol. 2013;15(2):334–346.[PMC free article] [PubMed] [Google Scholar]
- Rinaudi L., Fujishige N.A., Hirsch A.M., Banchio E., Zorreguieta A., Giordano W. Effects of nutritional and environmental conditions on Sinorhizobium meliloti biofilm formation. Res. Microbiol. 2006;157:867–875. [PubMed] [Google Scholar]
- Ruzicka F., Horka M., Hola V. Extracellular polysaccharides in microbial biofilm and their influence on the electrophoretic properties of microbial cells. In: Volpi N., editor. Capillary Electrophoresis of Carbohydrates: From Monosaccharides to Complex Polysaccharides. Springer; New York: 2011. pp. 105–126. [Google Scholar]
- Saini R., Saini S., Sharma S. Biofilm: a dental microbial infection. J. Nat. Sci. Biol. Med. 2011;2(1):71–75.[PMC free article] [PubMed] [Google Scholar]
- Sihorkar V., Vyas S.P. Biofilm consortia on biomedical and biological surfaces: delivery and targeting strategies. Pharm. Res. 2001;18(9):1247–1254. [PubMed] [Google Scholar]
- Socransky S.S., Haffajee A.D. Dental biofilms: difficult therapeutic targets. Periodontology. 2002;28:12–55. [PubMed] [Google Scholar]
- Stewart P.S., Costerton J.W. Antibiotic resistance of bacteria in biofilms. Lancet. 2001;358:135–138. [PubMed] [Google Scholar]
- ten Cate J.M. Novel anticaries and remineralizing agents: prospects for the future. J. Dent. Res. 2012;91(9):813–815. [PubMed] [Google Scholar]
- Tendolkar P.M., Baghdayan A.S., Gilmore M.S., Shankar N. Enterococcal surface protein, Esp, enhances biofilm formation by Enterococcus faecalis. Infect. Immun. 2004;72(10):6032–6039.[PMC free article] [PubMed] [Google Scholar]
- Tetz G.V., Artemenko N.K., Tetz V.V. Effect of DNase and antibiotics on biofilm characteristics. Antimicrob. Agents Chemother. 2009;53(3):1204–1209.[PMC free article] [PubMed] [Google Scholar]
- Trautner B.W., Darouiche R.O. Role of biofilm in catheter-associated urinary tract infection. Am. J. Infect. Control. 2004;32(3):177–183.[PMC free article] [PubMed] [Google Scholar]
- Villa F., Cappitelli F. Plant-derived bioactive compounds at sub-lethal concentrations: towards smart biocide-free antibiofilm strategies. Phytochem. Rev. 2013;12:245–254.[Google Scholar]
- Wilbur W.J., Lipman D.J. Rapid similarity searches of nucleic acid and protein data banks. Proc. Natl. Acad. Sci. U.S.A. 1983;80(3):726–730.[PMC free article] [PubMed] [Google Scholar]
- Xiao J., Klein M.I., Falsetta M.L., Lu B., Delahunty C.M., Yates J.R., Heydorn A., Koo H. The exopolysaccharide matrix modulates the interaction between 3D architecture and virulence of a mixed-species oral biofilm. PLoS Pathog. 2012;8(4):e1002623.[PMC free article] [PubMed] [Google Scholar]
Articles from Saudi Journal of Biological Sciences are provided here courtesy of Elsevier
